platinum
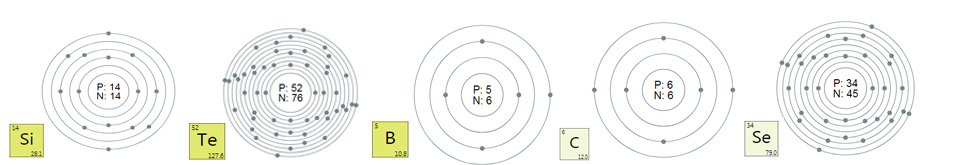
Platinum is a mineral, natural Pt from the platinum
group of the class of native elements, usually contains Pd, Ir, Fe, Ni.
Pure platinum is very rare, most of the samples are represented by the
ferrous variety (polyxene), and often by intermetallic compounds:
isoferroplatinum (Pt, Fe) 3Fe and tetraferroplatinum (Pt, Fe) Fe.
Platinum, represented by polyxene, is the most abundant platinum-group
mineral in the earth's crust.
Polyxene color ranges from silver white to steel black. The line is
metallic steel gray. Typical metallic luster. The reflectivity in
polished thin sections is high - 65-70.
Hardness 4-4.5, in iridium-rich varieties - up to 6-7. Possesses
malleability. The fracture is hooked. Cleavage is usually absent. Ud.
weight-15-19. The relationship between the reduced specific gravity and
the presence of voids occupied by natural gases, as well as inclusions
of foreign minerals, was noted. Magnetic, paramagnetic. Conducts
electricity well. Platinum is one of the most inert metals. It is
insoluble in acids and alkalis, with the exception of aqua regia.
Platinum also reacts directly with bromine, dissolving in it.
When heated, platinum becomes more reactive. It reacts with peroxides,
and upon contact with atmospheric oxygen, with alkalis. Thin platinum
wire burns in fluorine and produces a lot of heat. Reactions with other
non-metals (chlorine, sulfur, phosphorus) are less active. When heated
more strongly, platinum reacts with carbon and silicon, forming solid
solutions, similar to the metals of the iron group.
RESERVES AND PRODUCTION
Platinum Platinum is one of the rarest metals: its average content in
the earth's crust (clarke) is 5 × 10-7% by weight. Even the so-called
native platinum is an alloy containing 75 to 92 percent platinum, up to
20 percent iron, as well as iridium, palladium, rhodium, osmium, less
often copper and nickel.
Platinum compounds (mainly amminoplatinates) are used as cytostatics in
the treatment of various forms of cancer. Cisplatin (cis-dichlorodiammineplatinum
(II)) was the first to be introduced into clinical practice; however,
more effective carboxylate complexes of diammineplatinum - carboplatin
and oxaliplatin - are currently used.
Platinum and its alloys are widely used in jewelry making.
From the first quarter of the 19th century, it was used in World as an
alloying additive for the production of high-strength steels.
Platinum is used as a catalyst (most often in an alloy with rhodium, as
well as in the form of platinum black - a fine powder of platinum
obtained by reducing its compounds).
Platinum is used to make vessels and stirrers used in the melting of
optical glasses.
For the manufacture of chemically resistant and strong heating
laboratory glassware (crucibles, spoons, etc.).
For the manufacture of permanent magnets with high coercive force and
residual magnetization (alloy of three parts of platinum and one part of
cobalt PlK-78).
Special mirrors for laser technology.
For the manufacture of durable and stable electrical contacts in the
form of alloys with iridium, for example, contacts of electromagnetic
relays (alloys PLI-10, PLI-20, PLI-30).
Electroplated coatings.
Distillation retorts for hydrofluoric acid production, perchloric acid
production.
Electrodes for the production of perchlorates, perborates, percarbonates,
peroxoduseric acid (in fact, the use of platinum determines the entire
world production of hydrogen peroxide: electrolysis of sulfuric acid -
peroxoduseric acid - hydrolysis - distillation of hydrogen peroxide).
Insoluble anodes in electroplating.
Heating elements of resistance furnaces.
Manufacturing of resistance thermometers.
Coatings for microwave technology elements (waveguides, attenuators,
resonator elements).
.


Platinum is a mineral, natural Pt from the platinum
group of the class of native elements, usually contains Pd, Ir, Fe, Ni.
Pure platinum is very rare, most of the samples are represented by the
ferrous variety (polyxene), and often by intermetallic compounds:
isoferroplatinum (Pt, Fe) 3Fe and tetraferroplatinum (Pt, Fe) Fe.
Platinum, represented by polyxene, is the most abundant platinum-group
mineral in the earth's crust.
Polyxene color ranges from silver white to steel black. The line is
metallic steel gray. Typical metallic luster. The reflectivity in
polished thin sections is high - 65-70.
Hardness 4-4.5, in iridium-rich varieties - up to 6-7. Possesses
malleability. The fracture is hooked. Cleavage is usually absent. Ud.
weight-15-19. The relationship between the reduced specific gravity and
the presence of voids occupied by natural gases, as well as inclusions
of foreign minerals, was noted. Magnetic, paramagnetic. Conducts
electricity well. Platinum is one of the most inert metals. It is
insoluble in acids and alkalis, with the exception of aqua regia.
Platinum also reacts directly with bromine, dissolving in it.
When heated, platinum becomes more reactive. It reacts with peroxides,
and upon contact with atmospheric oxygen, with alkalis. Thin platinum
wire burns in fluorine and produces a lot of heat. Reactions with other
non-metals (chlorine, sulfur, phosphorus) are less active. When heated
more strongly, platinum reacts with carbon and silicon, forming solid
solutions, similar to the metals of the iron group.
RESERVES AND PRODUCTION
Platinum Platinum is one of the rarest metals: its average content in
the earth's crust (clarke) is 5 × 10-7% by weight. Even the so-called
native platinum is an alloy containing 75 to 92 percent platinum, up to
20 percent iron, as well as iridium, palladium, rhodium, osmium, less
often copper and nickel.
Platinum compounds (mainly amminoplatinates) are used as cytostatics in
the treatment of various forms of cancer. Cisplatin (cis-dichlorodiammineplatinum
(II)) was the first to be introduced into clinical practice; however,
more effective carboxylate complexes of diammineplatinum - carboplatin
and oxaliplatin - are currently used.
Platinum and its alloys are widely used in jewelry making.
From the first quarter of the 19th century, it was used in World as an
alloying additive for the production of high-strength steels.
Platinum is used as a catalyst (most often in an alloy with rhodium, as
well as in the form of platinum black - a fine powder of platinum
obtained by reducing its compounds).
Platinum is used to make vessels and stirrers used in the melting of
optical glasses.
For the manufacture of chemically resistant and strong heating
laboratory glassware (crucibles, spoons, etc.).
For the manufacture of permanent magnets with high coercive force and
residual magnetization (alloy of three parts of platinum and one part of
cobalt PlK-78).
Special mirrors for laser technology.
For the manufacture of durable and stable electrical contacts in the
form of alloys with iridium, for example, contacts of electromagnetic
relays (alloys PLI-10, PLI-20, PLI-30).
Electroplated coatings.
Distillation retorts for hydrofluoric acid production, perchloric acid
production.
Electrodes for the production of perchlorates, perborates, percarbonates,
peroxoduseric acid (in fact, the use of platinum determines the entire
world production of hydrogen peroxide: electrolysis of sulfuric acid -
peroxoduseric acid - hydrolysis - distillation of hydrogen peroxide).
Insoluble anodes in electroplating.
Heating elements of resistance furnaces.
Manufacturing of resistance thermometers.
Coatings for microwave technology elements (waveguides, attenuators,
resonator elements).
.

|

|